CUHK
News Centre
CUHK reveals how Helicobacter pylori ‘smuggles’ a toxic metal for its survival
A research team led by Professor Wong Kam-bo, Director and Professor in the School of Life Sciences at The Chinese University of Hong Kong (CUHK), in collaboration with Professor Susan Lea of the University of Oxford and the US National Cancer Institute, has revealed how Helicobacter pylori (H. pylori) survives in the human stomach by using a toxic metal, nickel, to activate urease, an enzyme that is essential for its survival in the acidic environment. This discovery can help develop novel drugs to counter H. pylori infection. The findings have been recently published in Science Advances, a prestigious international scientific journal.
H. pylori, which infects half of the human population and causes peptic ulcers worldwide, is the only pathogen that can colonise the human stomach. The bacterium produces large amount of urease, which converts urea, naturally found in the stomach, into ammonia that helps the pathogen to neutralise gastric acidity. However, urease requires a toxic nickel ion to function. The bacterium must find a way to “smuggle” the nickel ions into the urease without causing toxicity to itself.
In 2017, Professor Wong and his team showed that nickel ions are delivered from one helper protein to another through specific protein interactions. “Using a toxic metal for survival is not an easy task. H. pylori has evolved helper proteins to carry the nickel ions so that the toxic ions do not escape to the cell to cause problems,” he said. Nickel ions are present in trace amounts in the stomach. After uptake by transporter proteins on the surface of the bacterium, nickel ions are carried by the helper proteins to the urease.
The mystery of how nickel ions get inside the urease
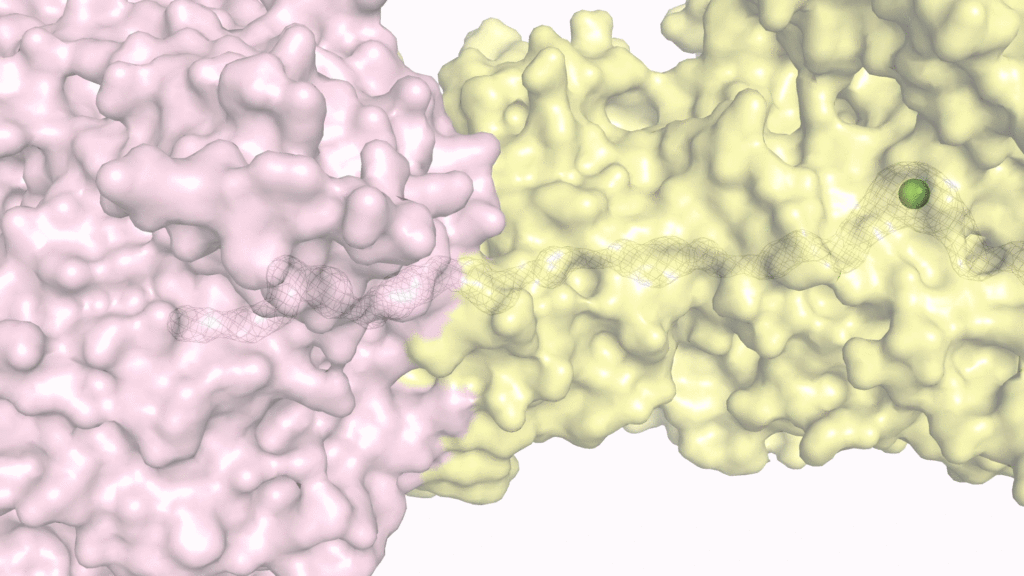
“Scientists have been puzzling how helper proteins give their nickel ions to the urease in the final step of urease activation. The problem was: the nickel binding pocket is located deep inside the urease protein. If the pocket is closed, how can the nickel ions get inside?” said Professor Wong. The research team solved the puzzle by using an electron microscope to visualise how the helper proteins interact with and deliver the toxic nickel ions to the urease. They showed that binding of the helper proteins to urease opens a tunnel inside the protein complex. As a result, the nickel ions can pass through this tunnel and get inside the urease. Since the nickel ions are delivered in a tunnel within the protein complex, they cannot escape to the bacterial cell to induce toxicity.
“The binding of the helper proteins serves as a key to open a secret passage that allows the toxic nickel ions to be smuggled to the active site of the urease. Without the key, the nickel binding pocket remains closed and the urease cannot be activated,” explained Professor Wong. “Our research opens up a new strategy to inhibit H. pylori infection. If we can find ways to block the delivery of nickel ions to the urease, H. pylori can no longer produce active urease and survive in the human stomach.”
The full text of the research paper can be found at:
https://www.science.org/doi/10.1126/sciadv.adf7790
Professor Wong and his team’s research in 2017 can be found at:
https://www.cpr.cuhk.edu.hk/en/press/cuhk-uncovers-how-helicobacter-pylori-uses-a-toxic-substance-to-keep-alive-in-human-stomach/