CUHK
News Centre
CUHK Unveils Novel Mechanism of Neuron DevelopmentSetting New Therapeutic Direction for Cerebellar Ataxia
A novel mechanism for how innate genetic factors modulate specific neurons to acquire their specific cell morphology to perform their normal functions has recently been revealed by Prof. Kin-ming KWAN from the School of Life Sciences at The Chinese University of Hong Kong (CUHK), together with his research team and collaborators. This work provides new insight into the molecular pathology of cerebellar ataxia and has been published recently in the leading international scientific journal Nature Communications.
Cerebellar ataxia is a rare neurological disorder characterized by tremor, loss of motor coordination and body balance. Prevalence of genetically inherited ataxia is estimated to be approximately 1 to 100 cases per million of the population worldwide. In Hong Kong, around 300 people have been diagnosed with spinocerebellar ataxia. The majority of the cerebellar ataxia are related to defective Purkinje cell development in the cerebellum. The cerebellum serves as the motor coordination centre in the central nervous system. In the cerebellum, Purkinje cells receive numerous signals from different inputs through their extensively branched dendrites. Defects in dendritic development of Purkinje cells thus disrupt the cerebellar circuitry and cause motor deficits. Therefore, investigating how Purkinje cell development is genetically regulated is a key to understanding the molecular pathology of cerebellar ataxia.
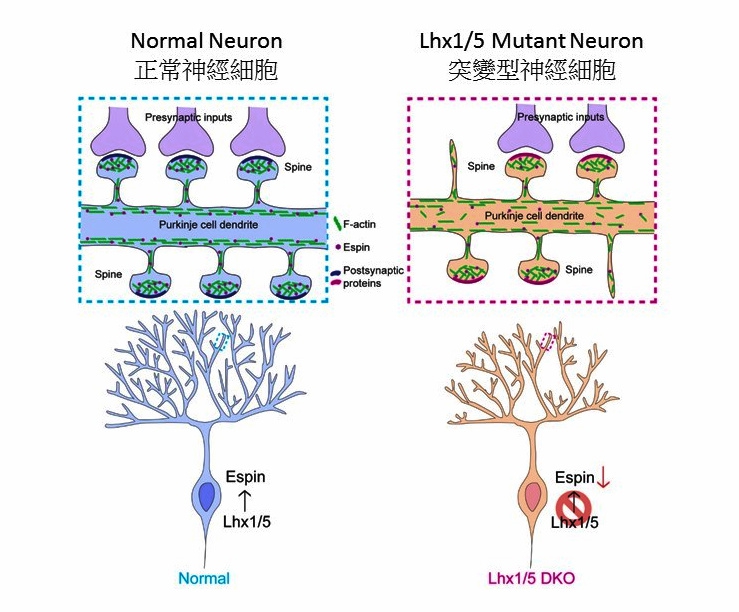
In this study, Professor Kwan and his team identified LIM-homeodomain transcription factors Lhx1 and Lhx5 as playing a critical role in regulating the dendritic development of Purkinje cells. Mutant mice lacking Lhx1 and Lhx5 in postnatal Purkinje cells display ataxic phenotypes due to dendritic defects found in the Purkinje cells. Their findings show that Lhx1 and Lhx5 can transcriptionally activate an F-actin regulatory protein, Espin, which in turns controls F-actin localization in the Purkinje cell dendrites. Proper F-actin localization facilitates the elongation and branching of dendrites and the maturation of dendritic spines. Moreover, they demonstrated that overexpression of Espin could rescue the dendritic defects of Purkinje cells in the mutant mice in vitro.
‘This is the first report showing that transcriptional factors can directly regulate an F-actin regulatory element to control cytoskeleton organization in Purkinje cells,’ Professor Kwan said, ‘Our findings provide a new direction in which upregulating specific F-actin regulatory elements in Purkinje cells can be a potential therapeutic intervention for cerebellar ataxia. We will next try to overexpress Espin in the cerebellum of mice in vivo to test whether it can relieve the ataxia symptoms in the live mice.’
This study was done in collaboration with Prof. Liwen JIANG from CUHK School of Life Sciences; Prof. Chi-chiu WANG from CUHK Department of Obstetrics and Gynecology; Prof. Wing-ho YUNG from CUHK School of Biomedical Sciences and Prof Jian-Dong HUANGfrom The University of Hong Kong. This project was supported by the General Research Fund, Collaborative Research Fund and Areas of Excellence (AoE) Scheme of the Hong Kong Research Grants Council, Health and Medical Research Fund of Food and Health Bureau HKSAR, as well as the Centre for Cell and Developmental Biology and the Partner State Key Laboratory of Agrobiotechnology of CUHK.
Brief biography of Prof. Kin-ming Kwan
Prof. Kin-ming Kwan is Associate Professor and Director of the Natural Sciences Programme in the Faculty of Science at CUHK. He received undergraduate and doctoral training at The University of Hong Kong, and postdoctoral training at The University of Texas MD Anderson Cancer Center (US). Since 2006, Professor Kwan has been investigating the genetic regulation of neuronal cell development in cerebellum and its relationship to cerebellar ataxia disease conditions. He joined CUHK in 2006 and is recipient of the CUHK Young Researcher Award, 2008 and the CUHK Science Faculty Teaching Award, 2009 and 2013.
Prof. Kin-ming KWAN (left) and Miss Nga Chu LUI of School of Life Sciences at CUHK shed new insights into the molecular pathology of cerebellar ataxia.