CUHK
News Centre
CUHK Introduces Nasal Strip Sampling for COVID-19 Test As a Superior Tool for Surveillance in a Wide Age-Range Population
A research team from the Faculty of Medicine at The Chinese University of Hong Kong (CU Medicine) has recently introduced the nasal strip test as a novel and sensitive method to detect SARS-CoV-2. The team recruited approximately 40 hospitalised COVID-19 patients, aged 6 to 74, and obtained their nasal epithelial lining fluid by nasal strip. The samples were then compared against standard sampling methods including pooled nasopharyngeal and throat swabs or deep throat saliva.
Results showed that accuracy of nasal strip test for SARS-CoV-2 is comparable to standard tests and even out-performed saliva specimens, especially in paediatric subjects. Specimens collected by nasal strip are also stable at room temperature for 72 hours. These results suggest that the nasal strip is a superior tool for community-based surveillance testing since it is a low-risk method which can be easy to administer in subjects of a wide age-range. Study details have been published in the Journal of Infection.
Standard methods pose disease transmission risk or inconsistency in sample quality
COVID-19 has caused over 7,000 laboratory confirmed cases in Hong Kong. Collecting nasopharyngeal/oropharyngeal specimens by swabs for detection of this novel coronavirus is one of the standard methods. However, not only does this require trained-personnel to perform, the process can easily trigger sneezing, and coughing, which can pose a risk of disease transmission to surrounding people.
The deep throat saliva test is another option for sampling. Although this method can be self-administered by the users, it did not translate well to all individuals in the community setting. Dr. Kate Ching Ching CHAN, Assistant Professor of the Department of Paediatrics at CU Medicine, explained, “Young children and the elderly are less able to provide deep throat saliva samples as the collection technique is particularly difficult for them. This difficulty results in a high variability of sample quality which in turn reduces test sensitivity. This is not ideal for community surveillance.”
In view of this, the Department of Paediatrics at CU Medicine introduced the nasal strip as a novel collection method and assessed its application in both paediatric and adult subjects. 38 asymptomatic and symptomatic subjects from the Prince of Wales Hospital were recruited, among which there were 20 infected adults (22 to 74 years old) and 18 infected children/adolescents (6 to 17 years old).
Accuracy of nasal strip test was comparable to standard tests and out-performed saliva specimen in paediatric subjects
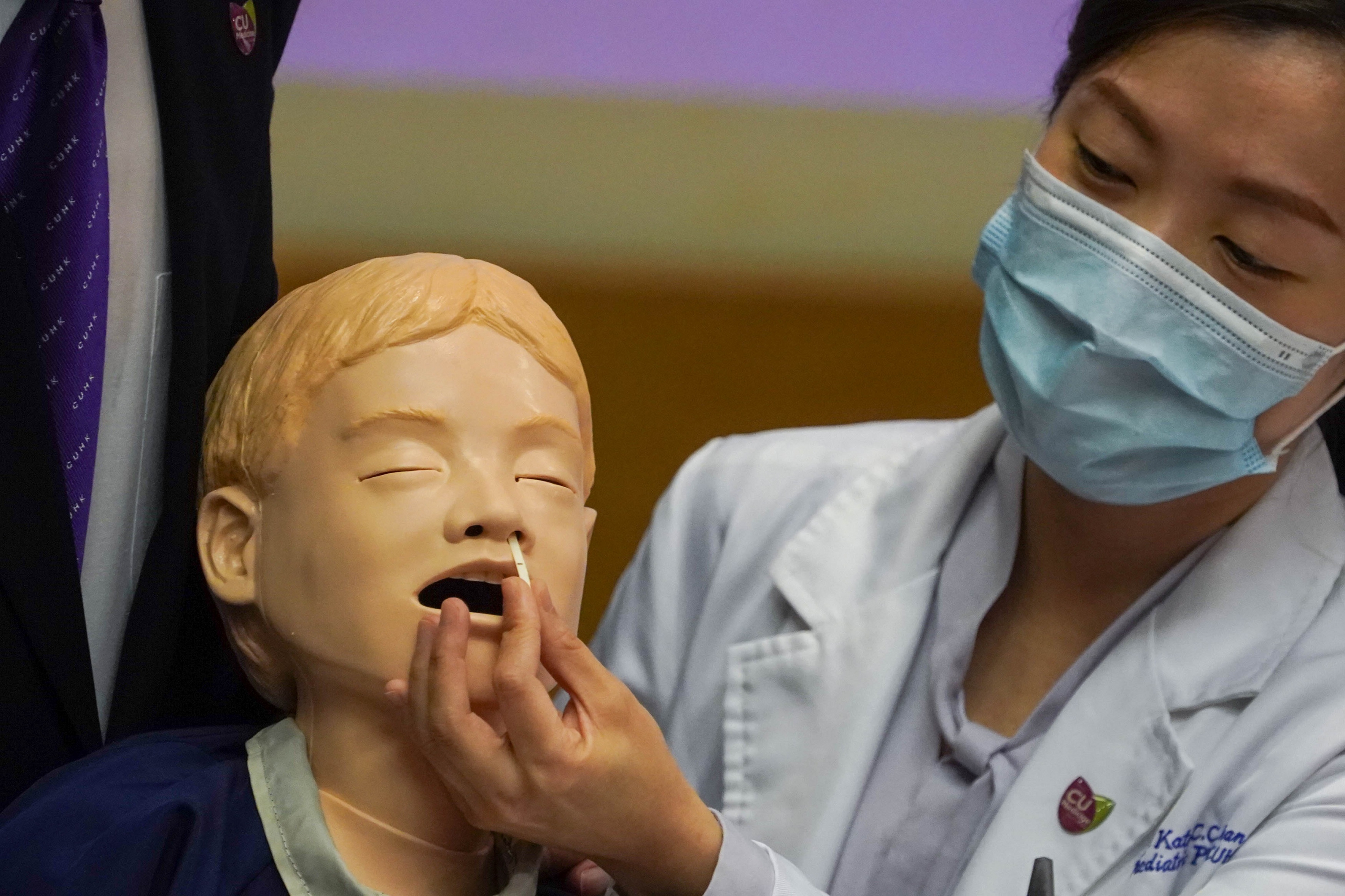
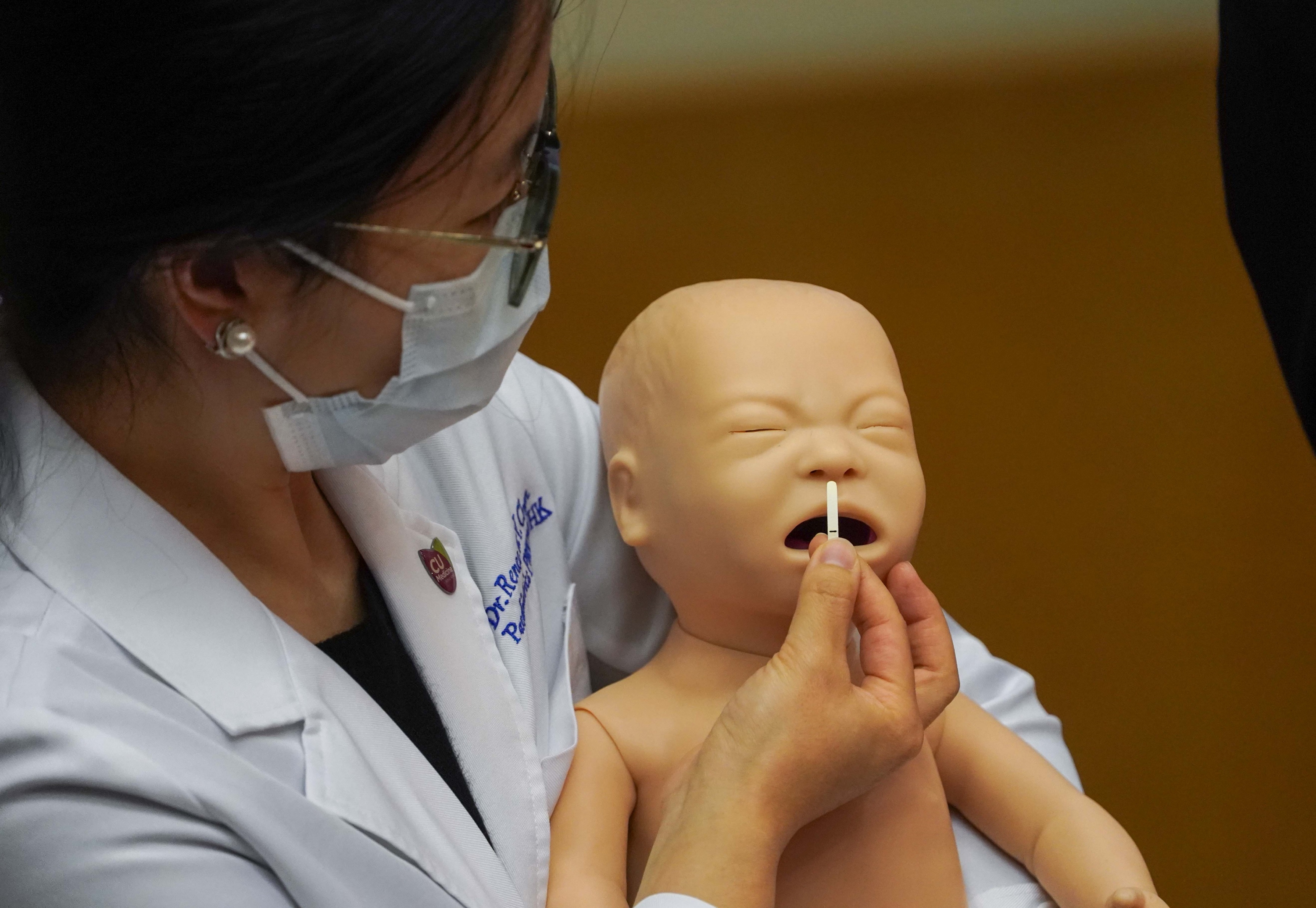
Nasal epithelial lining fluid of the subjects was collected by nasal strips, which were prepared and cut into a designated size by the research team. The specimens were then compared against pooled nasopharyngeal and throat swabs or deep throat saliva collected from the same subject group within 24 hours after the nasal strip test. 13 paired nasal swabs were also collected by a healthcare worker right before the collection of the nasal strips to evaluate their detection performance. All samples were subjected to viral RNA quantitation by real-time PCR.
Results are listed as below:
- Compared with nasopharyngeal and throat swabs, nasal strip sampling achieved a result agreement of 95.2%. This shows the 2 sampling methods share similar accuracy.
- The result agreement of nasal strip sampling reduced to 68.2% when compared with deep throat saliva. However, nasal strip out-performed deep throat saliva in 6 occasions out of the 7 discrepant samples identified, where negative result was reported in the latter. Four of these deep throat saliva specimens were collected from paediatric patients.
- There were no significant differences detected between cycle threshold (Ct) values of the paired samples of nasal strip and nasal swab, which reflects that the amount of virus detected through the two sampling methods is similar.
- Nasal strip pairs were collected from 6 patients to determine viral stability over time and viral RNA remained detectable after storage at room temperature for 72 hours.
First author of the study, Dr. Renee Wan Yi CHAN, Assistant Professor of the Department of Paediatrics at CU Medicine, stated, “The performance of the nasal strip is likely the result of steady nasal epithelial lining fluid absorption into the strip which is in close contact with the nasal mucosa. This reduces sample variability. Our results suggest that the nasal strip test would be superior to the deep throat saliva test for surveillance purposes over paediatric and elderly populations as the latter shows reduced sensitivity in these populations.”
The nasal strip test is also a less uncomfortable collection method with lower risk than nasopharyngeal and throat swabs as it is less traumatic and irritating. The application of the nasal strip reduces the possibility of any sneezes and coughs and therefore lessens the risk of virus transmission.
Professor Hugh Simon Hung San LAM, Professor of the Department of Paediatrics at CU Medicine, concluded, “In addition to its accuracy and lower risk of virus transmission, the stability of nasal strip samples at room temperature potentially allows the specimens to be posted to the laboratory which usually takes at least a day. All of these suggest that nasal strip testing can serve an important role in community-based surveillance programmes especially in populations such as young children and elderly adults where standard methods are more likely to fail.”
Study details have just been published in the Journal of Infection: https://doi.org/10.1016/j.jinf.2020.11.009.
CU Medicine has recently introduced a novel nasal strip test as a sensitive and accurate method to detect SARS-CoV-2. It could be a superior tool for community based surveillance test since it is a simple, less irritative and low risk method which is easy to administer in subjects of a wide age range. From left: Dr. Renee CHAN, Assistant Professor; Prof. Simon LAM, Professor; Prof. Albert LI, Chairman; and Dr. Kate CHAN, Assistant Professor, the Department of Paediatrics at CU Medicine.
The nasal strips used in the study conducted by CU Medicine are customized in different sizes to fit for children at different ages to detect SARS-CoV-2.
The nasal strips used in the study conducted by CU Medicine are customized in different sizes to fit for children at different ages to detect SARS-CoV-2.