CUHK
News Centre
CUHK Finds Link Between ‘Fuz’ Protein Level and Neurological DisordersNew Treatment Paths for Neural Diseases
Professor H.Y. Edwin Chan and his research team from the School of Life Sciences of The Chinese University of Hong Kong (CUHK) have recently elucidated a novel pathogenic mechanism for a group of rare genetic neuronal disorders, known as polyglutamine (polyQ) diseases, including spinocerebellar ataxias and Huntington’s disease. This work has been published in the prestigious scientific journal, EMBO Reports, in which all areas of molecular biology research and expert reviews are covered.
Spinocerebellar ataxia type 3/Machado-Joseph disease (SCA3/MJD) is the most common form of dominantly inherited ataxia in many populations, including Hong Kong. SCA3/MJD patients often present problems with gait and balance, blurred vision, and speech difficulties. The symptoms are progressive, and to date SCA3/MJD remains an incurable disease.
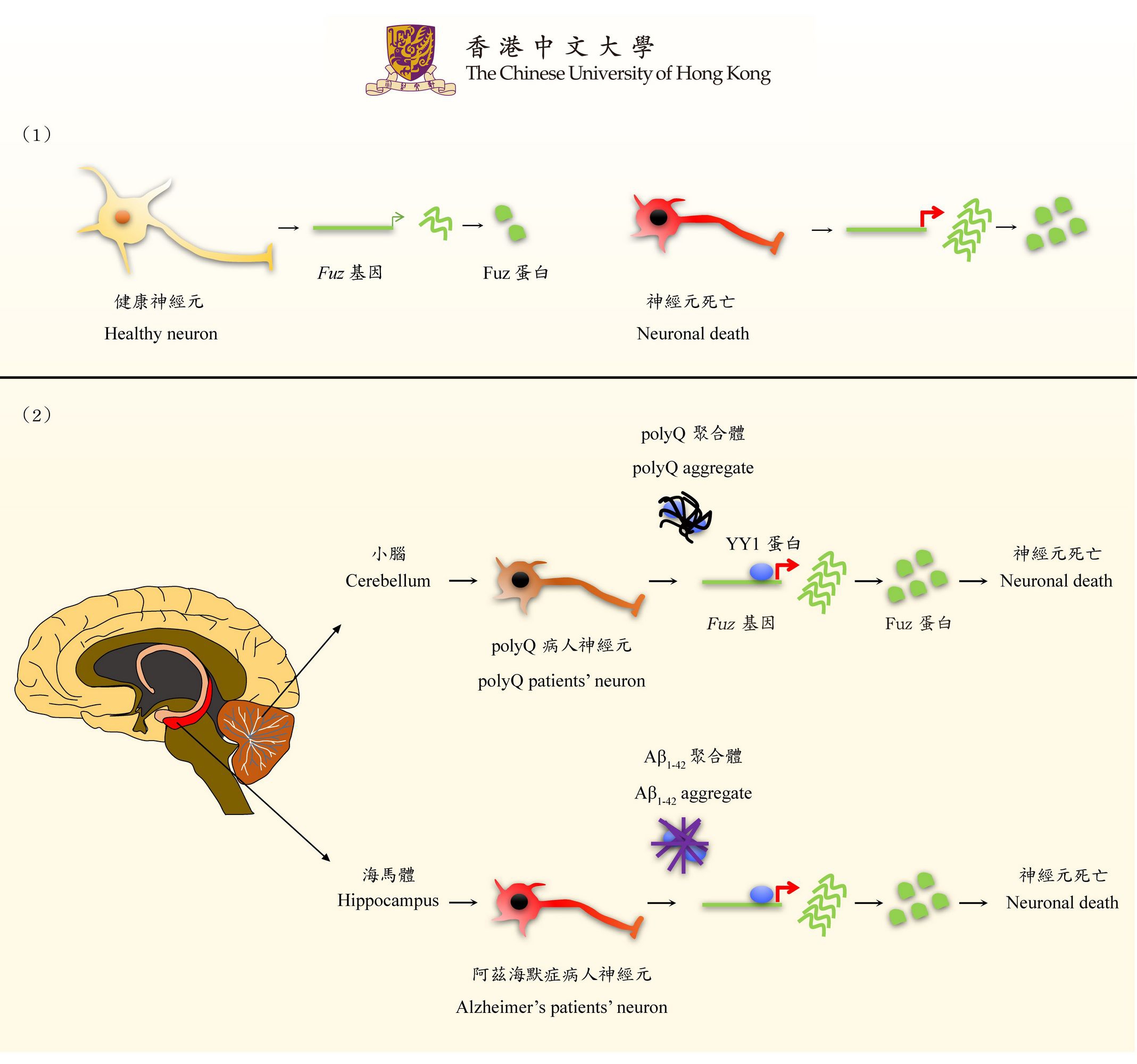
Misfolded protein may contribute to the pathogenesis of neurological disorders
Proteins are the major work force in every single cell type, including neurons, in the human body. Proteins need to adopt proper functional shapes before they can carry out their duties in cells. Any improper folding of a protein may result in cellular malfunctioning and lead to diseases. These malformed faulty proteins eventually form macroscopic aggregates that interfere with the functionality of other normal proteins in the same cell. Misfolded disease proteins (polyQ protein aggregates) are detected in the deteriorating neurons of SCA3/MJD and other polyQ disease patients.
Correlations between YY1, Fuz and polyQ diseases revealed for the first time
Professor Chan and his team showed that the SCA3/MJD polyQ protein aggregates perturb the function of Yin Yang 1 protein (YY1). In neurons, YY1 functions as a repressor of Fuz protein (Fuz). When the function of YY1 is compromised, the expression level of Fuz will increase. Once it exceeds 2.5 times the normal level, it will trigger apoptosis and cause cell death in neurons. The team further detected an abnormal induction of Fuz expression in SCA3/MJD patients, proving that this is a pathogenic mechanism of polyQ diseases. This is the first time that the new function of Fuz in inducing neuronal death has been discovered, and also the first report to demonstrate the role of YY1 and Fuz in polyQ diseases.
Overexpressed Fuz also presents in common neurodegenerative diseases such as Alzheimer’s disease
Professor Chan said, ‘In the past, scientists only considered Fuz as a player in neurodevelopment. We are the first to report this unnoticed cell death-inducing function of Fuz and unveil its role in polyQ diseases. Intriguingly, we also observed that in Alzheimer’s disease, the expression level of Fuz also increased. This unexpected and exciting research finding tells us the Fuz-mediated apoptosis pathway plays some common roles in neurological disorders in general. We are now actively investigating the involvement of Fuz in other disease conditions, and finding compounds that can bring Fuz upregulation back down to a normal level. This will be a novel therapeutic direction toward polyQ and Alzheimer’s diseases. It is also my hope that our discovery can demonstrate how rare disease research can make contributions to the advancement of biomedical science research in neural disease biology.’
Professor Edwin Chan and international scholars work together to find the causes of rare diseases and push forward treatment
Since 1998, Professor Edwin Chan has devoted himself to the study of rare neurological and neuromuscular diseases. When Professor Chan served as a visiting scholar at Churchill College, Cambridge in 2014, he conducted a new cerebellar atrophy study at Cambridge’s Department of Genetics. He then established a research collaboration network for spinocerebellar ataxias and Huntington’s disease, and has been running a multidisciplinary research programme with partners from Denmark, France, Italy, Poland, the UK and the US, hoping to find more about the causes of rare diseases, thus developing treatment plans.
This work was supported by the CUHK Gerald Choa Neuroscience Centre, the General Research Fund of the Hong Kong Research Grants Council, the Chow Tai Fook Charity Foundation, and the Hong Kong Spinocerebellar Ataxia Association.
Brief biography of Professor H.Y. Edwin Chan
Professor H.Y. Edwin Chan is Professor and Director of the Master of Science Degree Programme in Biochemical and Biomedical Sciences in the School of Life Sciences. After completing his bachelor’s degree in biochemistry at CUHK, he was funded by the Croucher Foundation and went to The University of Cambridge to take a PhD in genetics. Then he was supported by the Wellcome Trust in the UK to receive post-doctoral training in biomedicine at The University of Pennsylvania.
Professor Chan participates in editorial services for academic journals and international funding agencies. He actively participates in community services, including being the chairperson of the scientific and medical consultative committee of the Hong Kong Spinocerebellar Ataxia Association, and an advisor to the Hong Kong Alliance for Rare Diseases.
Original article: Planar cell polarity gene Fuz triggers apoptosis in neurodegenerative disease models, EMBO Reports (2018) http://m.embor.embopress.org/content/early/2018/07/18/embr.201745409
This finding demonstrates the function of Fuz in inducing neuronal death. The study of this function in polyQ disorders will advance the mechanistic research in common neurological diseases.
Professor Edwin Chan (left) and Dr Stephen Chen of CUHK School of Life Sciences point out that overexpressed Fuz also presents in common neurodegenerative diseases such as Alzheimer's disease