CUHK
News Centre
Breakthrough in Energy Storage TechnologyCUHK Develops Aqueous Redox Flow Battery with Record Energy Density
A high-energy-density zinc/iodine-bromide redox flow battery (ZIBB) has recently been developed by Prof. Yi-Chun Lu, Assistant Professor of the Department of Mechanical and Automation Engineering, The Chinese University of Hong Kong and her research team. ZIBB achieved the highest reported energy density for aqueous redox flow batteries to-date. The breakthrough was published in the renowned journal Energy & Environmental Science in early 2017, and was recently featured by the magazine Chemistry World, published by The Royal Society of Chemistry, United Kingdom.
Bromide ion (Br-): The Key to Releasing Energy Density
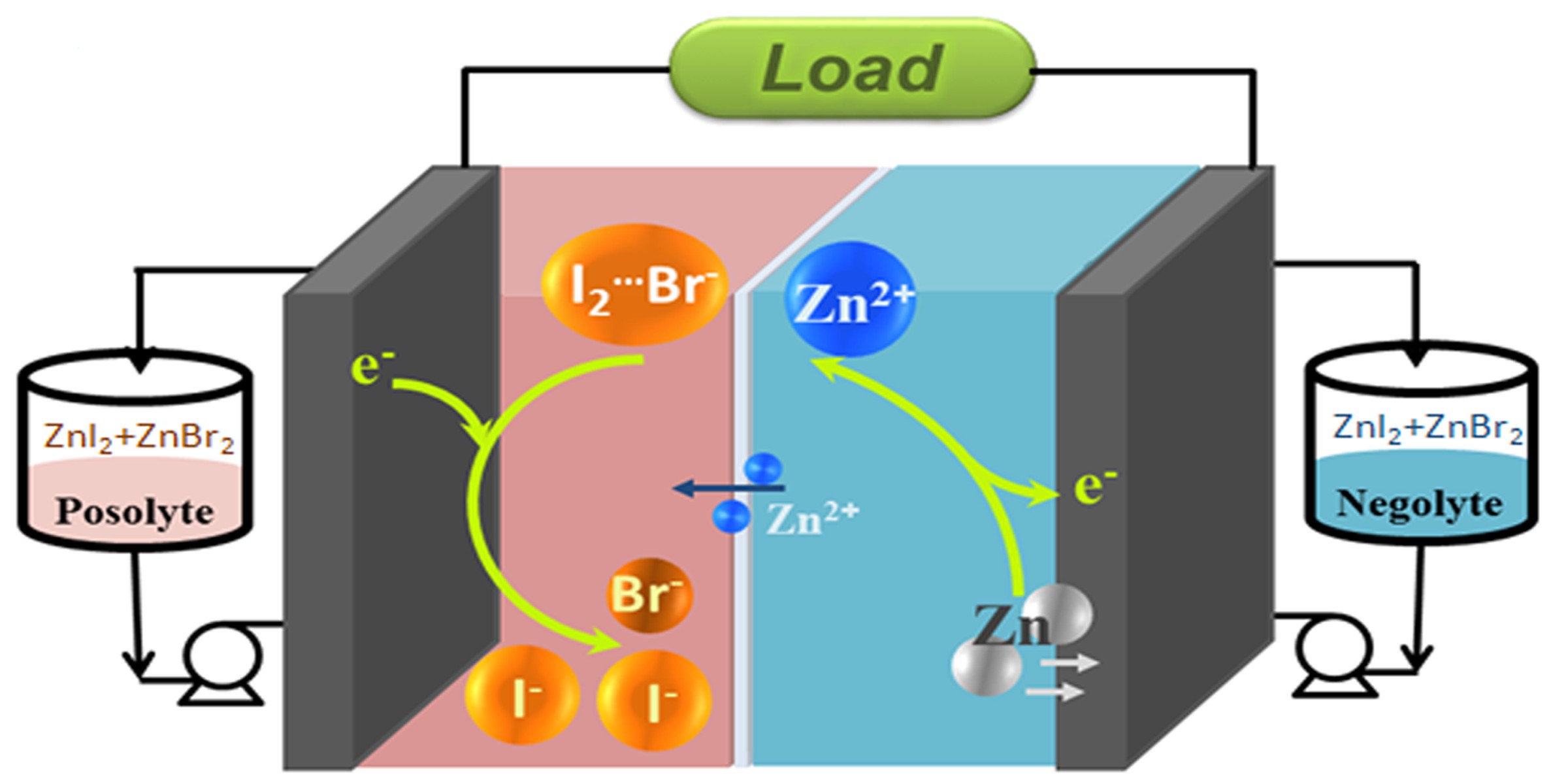
Aqueous Redox Flow Battery (RFB) is a device that generates electricity by electron transfer between two electrolytes. RFB is safe, friendly to the environment, with high design flexibility and long life (several decades), and it appears to have a high commercialization potential. With the introduction of Bromide ions (Br-), Professor Lu’s research group boosted the energy density of ZIBB to as high as 101 Wh L-1, achieving the highest reported energy density to-date, i.e. an improvement of at least 20% in capacity relative to a control system.
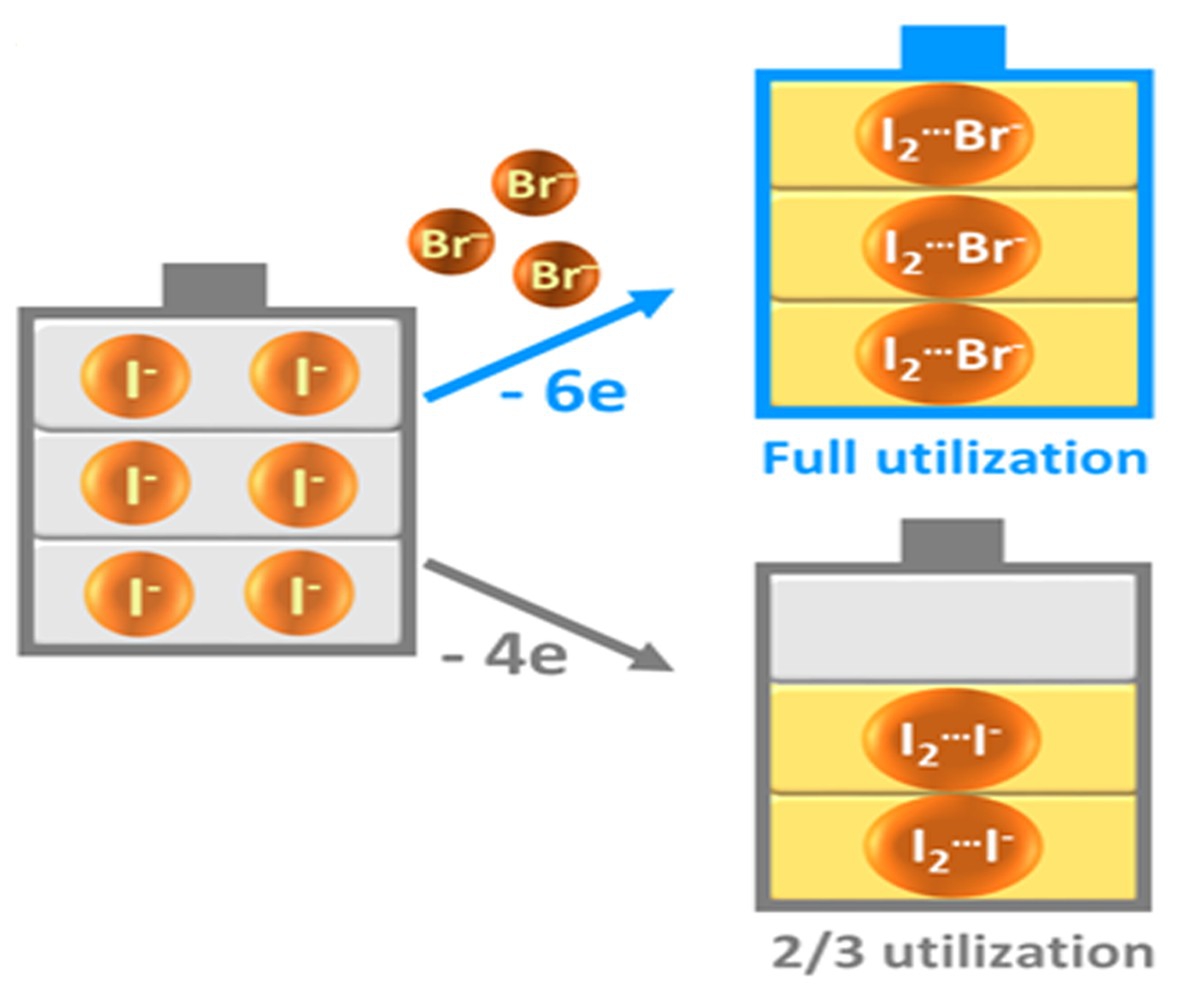
In zinc/iodine RFBs, highly soluble zine iodide is the major active material in the electrolyte, with iodide ions (I-) and zinc (Zn) being the electrochemical active ingredients at the positive and negative electrodes respectively. Free iodine (I2) is present in the battery, and iodide ions (I-) act as the stabilizing agent to form triiodide ions (I3-), thereby stabilizing the cycle life of the battery (efficiency as high as 95% over 50 cycles). However, the power of the iodide ions (I-) in contributing battery capacity is wasted as they are “trapped” as a stabilizing agent.
The group therefore pioneered the innovation by which bromide ions are introduced as a replacement for the “trapped” iodide ions (I-), i.e. forming iodine bromide ions (I2Br–) by reacting bromide ions (Br-) with iodine (I2). The process still allows a stable cycle life in the battery, without sacrificing energy capacity.
The Potential of ZIBB
This new energy storage system with high energy density and a stable cycle life has potential in the growing market for electric cars. “The price of electric cars would be significantly lowered if this type of battery was adopted, with much longer driven mileage. Moreover, this type of battery is much safer as in a regular crash. Some work in commercializing the technology is underway. The technology is promising when applied to higher performing, lower cost, and larger scale energy storage systems.” Professor Lu commented.
Their experiment also proved that the technology is able to boost the theoretical energy density of all electrochemical energy storage systems that involve iodine by one third. In the future, the team is going to research further into low-cost and high-performance electrode and membrane materials to optimize the technology.
About the Research Project “Smart Solar Energy Harvesting, Storage and Utilization”
The study is part of the 5-year research project “Smart Solar Energy Harvesting, Storage and Utilization” led by Prof. Ching-ping Wong, Dean of Engineering, CUHK. The project has been funded by the Theme-based Research Scheme (TRS) of the Research Grants Council (RGC) of the Hong Kong Government (HK$ 60.33 million) since 2014, with another HK$ 13.8 million from CUHK and HK$ 3 million from other partner universities. More than 30 scholars from CUHK, The Hong Kong Polytechnic University, The Hong Kong University of Science and Technology and The University of Hong Kong have been working together to enhance the efficiency of solar power and the penetration of the technology.
About Prof. Yi-Chun Lu
Prof. Yi-Chun Lu received her B.S. degree in Materials Science & Engineering from the National Tsing Hua University, Taiwan, in 2007. She received her Ph.D. degree in Materials Science & Engineering from the Massachusetts Institute of Technology (MIT), Cambridge, USA in 2012. Professor Lu has been appointed as a research affiliate of MIT since 2013. She is currently an Assistant Professor in the Department of Mechanical and Automation Engineering at CUHK. Professor Lu has been conferred various CUHK and international research and teaching awards, including the University Education Award, CUHK (2016), the Vice-Chancellor’s Exemplary Teaching Award, CUHK (2014), the Early Career Award, Research Grant Council, Hong Kong SAR (2014), Massachusetts Institute of Technology Martin Family Society of Fellows for Sustainability (2009) and the Taiwan National Science Council Outstanding Research Innovation Award (2007).
International ICT EXPO 2017
The research will be showcased in the “International ICT EXPO 2017” organized by the Hong Kong Trade Development Council from 13 – 16 April 2017 at the Hong Kong Convention and Exhibition Center at Wan Chai. More innovative research projects will be shown and all are welcome.
Date: 13 – 16 April 2017
Time: 9:30am – 6:30pm (Thursday – Saturday)
9:30am – 5:00pm (Sunday)
Venue: Hong Kong Convention and Exhibition Center, 1 Expo Drive, Wan Chai, Hong Kong (CUHK booth no.: B04 (Hall 3G))
The team is operating experiment work at a laboratory. (From left) Dr. Guo-Ming Weng, Prof. Yi-Chun Lu and Ms. Zhejun Li.
During the charging process, bromide (Br-) replaces iodide as the complexing agent to form iodine bromide ions, releasing the iodide (I-) to contribute energy capacity.